Chemo-, regio-, and stereo-selective perfluoroalkylations by a Grignard complex with zirconocene†
Abstract
The synthesis of highly reactive perfluoroalkyl Grignard reagents with early transition metal zirconocene complexes and their new types of highly chemo-, regio-, and stereo-selective perfluoroalkylation reactions are reported with epoxides in particular. The zirconocene complex is advantageous in activating the perfluoroalkyl Grignard species. The zirconocene·Grignard complexes were clarified by DOSY. Both 1H and 19F DOSY analyses show that the addition of MAO and dioxane to the mixture of RFMgCl and Cp2ZrCl2 connects Cp2Zr and RFMg to generate the zirconocene/perfluoroalkyl-Grignard/dioxane complex.
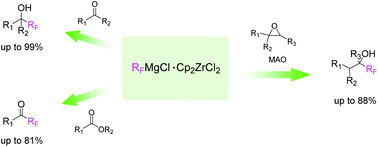
- This article is part of the themed collection: Fluorine

 Please wait while we load your content...
Please wait while we load your content...