Non-redox metal ions can promote Wacker-type oxidations even better than copper(ii): a new opportunity in catalyst design†
Abstract
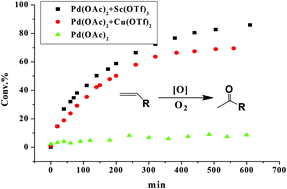
In Wacker oxidation and inspired Pd(II)/Cu(II)-catalyzed C–H activations, copper(II) is believed to serve in re-oxidizing of Pd(0) in the catalytic cycle. Herein we report that non-redox metal ions like Sc(III) can promote Wacker-type oxidations even better than Cu(II); both Sc(III) and Cu(II) can greatly promote Pd(II)-catalyzed olefin isomerization in which the redox properties of Cu(II) are not essential, indicating that the Lewis acid properties of Cu(II) can play a significant role in Pd(II)-catalyzed C–H activations in addition to its redox properties. Characterization of catalysts using UV-Vis and NMR indicated that adding Sc(OTf)3 to the acetonitrile solution of Pd(OAc)2 generates a new Pd(II)/Sc(III) bimetallic complex having a diacetate bridge which serves as the key active species for Wacker-type oxidation and olefin isomerization. Linkage of trivalent Sc(III) to the Pd(II) species makes it more electron-deficient, thus facilitating the coordination of olefin to the Pd(II) cation. Due to the improved electron transfer from olefin to the Pd(II) cation, it benefits the nucleophilic attack of water on the olefinic double bond, leading to efficient olefin oxidation. The presence of excess Sc(III) prevents the palladium(0) black formation, which has been rationalized by the formation of the Sc(III)⋯H–Pd(II) intermediate. This intermediate inhibits the reductive elimination of the H–Pd(II) bond, and facilitates the oxygen insertion to form the HOO–Pd(II) intermediate, and thus avoids the formation of the inactive palladium(0) black. The Lewis acid promoted Wacker-type oxidation and olefin isomerization demonstrated here may open up a new opportunity in catalyst design for versatile C–H activations.

 Please wait while we load your content...
Please wait while we load your content...