Synthesis and spectroscopic characterization of group 4 post-metallocenes bearing (σ-aryl)-2-phenolate-6-pyridyl and -isoquinolinyl auxiliaries†‡
Abstract
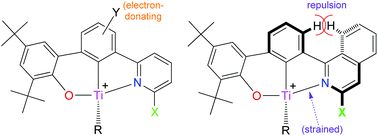
A new series of group 4 bis(benzyl) complexes supported by (σ-aryl)-2-phenolate-6-pyridyl [O,C,N] ligands have been prepared, and all derivatives have been characterized by multinuclear NMR spectroscopy. In the 1H NMR spectrum of the Ti derivative where [N] = (ortho-F)-substituted isoquinolinyl, one of the two CH2 resonances is observed as a doublet of doublets (collapsing to a normal d upon 19F-decoupling), whereas the [1H,19F]-HMQC correlation spectrum reveals a strong crosspeak for this dd resonance only, thus indicating the presence of intramolecular C–H⋯F–C interactions. [1H,19F]-HMBC experiments have been performed which reveal a significant scalar component for this coupling and confirm that the interactions are genuine. The contrasting NMR spectral patterns for the (ortho-F)-pyridyl Hf analogue, which exhibits two sets of non-identical doublet of doublets for the methylene resonances, have been rationalized. The activities of the isoquinolinyl-based Ti-[O,C,N] catalysts for ethylene polymerization are superior to those of pyridyl-based congeners.
- This article is part of the themed collection: In memory of Professor Kenneth Wade

 Please wait while we load your content...
Please wait while we load your content...