Preparation, characterization and properties of four new trivalent lanthanide complexes constructed using 2-bromine-5-methoxybenzoic acid and 1,10-phenanthroline†
Abstract
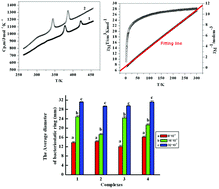
Four dinuclear trivalent lanthanide complexes of the general formula [Ln2(2-Br-5-MOBA)6(phen)2] (Ln = Nd(1), Sm(2), Ho(3), Er(4); 2-Br-5-MOBA = 2-bromine-5-methoxybenzoate, phen = 1,10-phenanthroline) were prepared and structurally characterized. The complexes 1 to 4 are isostructural with a coordination number of nine. The carboxylic acid ligands adopt bridging, bidentate chelating, and tridentate chelating bridging modes to coordinate with Ln(III) ions. The structures of the complexes were confirmed on the basis of elemental analysis, molar conductance, thermogravimetric analysis (TGA), and IR and UV spectra. The heat capacities and thermal circulating processes of the prepared complexes were performed on a differential scanning calorimeter under a nitrogen atmosphere. Two remarkable solid–solid phase transitions existed both in the heat capacities and thermal circulating processes. The bacteriostatic activities of the complexes were evaluated against Candida albicans, Escherichia coli and Staphylococcus aureus. Besides, the luminescent property of complex 2 was also investigated, and the magnetic properties of complexes 3 and 4 were also discussed in detail.

 Please wait while we load your content...
Please wait while we load your content...