The conjugates of ferrocene-1,1′-diamine and amino acids. A novel synthetic approach and conformational analysis†
Abstract
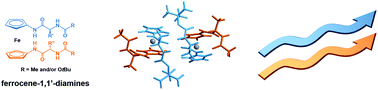
A novel synthetic approach toward a poorly explored bioorganometallic consisting of ferrocene-1,1′-diamine bearing structurally and chirally diverse amino acid sequences is reported. Until now, ferrocene-1,1′-diamine was suitable for accommodating only identical amino acid sequences at its N-termini, leading to the symmetrically disubstituted homochiral products stabilized through a 14-membered intramolecular hydrogen-bonded ring as is seen in antiparallel β-sheet peptides. The key step of the novel synthetic pathway is the transformation of Ac–Ala– –Fn–
–Fn– OH (5) (Fn = 1,1′-ferrocenylene) to orthogonally protected Ac–Ala–
OH (5) (Fn = 1,1′-ferrocenylene) to orthogonally protected Ac–Ala– –Fn–
–Fn– Boc (7). The spectroscopic analysis (IR, NMR, CD) of the novel compounds, corroborated with DFT studies, suggests the interesting feature of the ferrocene-1,1′-diamine scaffold. The same hydrogen-bonding pattern, i.e. a 14-membered hydrogen-bonded ring, was determined both in solution and in the solid state, thus making them promising, yet simple scaffolds capable of mimicking β-sheet peptides. In vitro screening of potential anticancer activity in Hep G2 human liver carcinoma cells and Hs 578 T human breast cancer cells revealed a cytotoxic pattern for novel compounds (150–500 μM) with significantly decreased cell proliferation.
Boc (7). The spectroscopic analysis (IR, NMR, CD) of the novel compounds, corroborated with DFT studies, suggests the interesting feature of the ferrocene-1,1′-diamine scaffold. The same hydrogen-bonding pattern, i.e. a 14-membered hydrogen-bonded ring, was determined both in solution and in the solid state, thus making them promising, yet simple scaffolds capable of mimicking β-sheet peptides. In vitro screening of potential anticancer activity in Hep G2 human liver carcinoma cells and Hs 578 T human breast cancer cells revealed a cytotoxic pattern for novel compounds (150–500 μM) with significantly decreased cell proliferation.

 Please wait while we load your content...
Please wait while we load your content...