Abstract
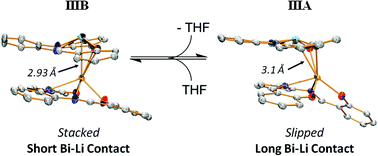
From the reaction between BiCl3 (1 equiv.) and LiN(SiMe3)2 (4 equiv.) and LH2 (2 equiv.), where L = a tetraphenylporphyrin, TPP, an octaethylporphyrin, OEP and phsalen in THF the title compounds have been obtained I LiBiTPP2, II LiBiOEP2, and IIIA LiBi(phsalen)2 and IIIB LiBi(phsalen)2·THF. Crystals grown from CH2Cl2–hexanes are colored; I (green), II (red-purple) and IIIA and IIIB (red-orange). The molecular structures of compound I, IIIA and IIIB were determined by single-crystal X-ray crystallography and are shown to have short Li⋯Bi bonds of distance 2.8 Å involving the LiL−BiL+. Compound IIIA shows a slipped structure involving Li to two oxygens and a Li⋯Bi distance of 3.1 Å. Compounds IIIA and IIIB undergo a rapid reversible exchange in toluene-d8 at 90 °C. The MALDI-MS yields weak molecular ions due to LiBiL2+/− with more intense ions due to BiL+ and LiL− in the positive and negative modes. The short Li+ to Bi3+ distances are comparable to those seen in Li⋯Bi compounds, such a LiBiR2, and are comparable to those seen by Pyykkö (P. Pyykkö, J. Phys. Chem. A, 2015, 119, 2326; P. Pyykkö and M. Atsumi, Chem. – Eur. J., 2009, 15, 186; P. Pyykkö and M. Atsumi, Chem. – Eur. J., 2009, 15, 12770) for Li–Bi bonds. These can be seen to be involving Bi6s6p hybrid lone-pairs to Li+ atoms. The lithium bis(bistrimethylsilyl)amide (2 equiv.) and phsalenH2 in THF gave a compound having Li2L·2THF, IV. Crystallographically compound IV contains two Li+ atoms, one coordinated to five atoms LiO2N2·THF. and the other being coordinated to three atoms, LiO2·THF. By 7Li and 1H NMR both lithium atoms share an equivalent environment.
- This article is part of the themed collection: In memory of Professor Kenneth Wade

 Please wait while we load your content...
Please wait while we load your content...