Synthesis, characterisation and properties of rare earth oxyselenides A4O4Se3 (A = Eu, Gd, Tb, Dy, Ho, Er, Yb and Y)†
Abstract
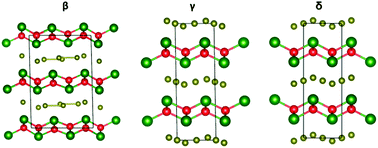
Rare earth oxyselenides A4O4Se3 (A = Nd, Sm, Eu, Gd, Tb, Dy, Ho, Er, Yb and Y) were synthesised using solid state reactions and three new structure types (β, γ, and δ) were observed. A4O4Se3 materials adopt either the α (A = Nd, Sm), β (A = Eu), γ (A = Gd, Tb) or δ (A = Dy, Ho, Er, Yb, Y) structure depending on the rare earth radius. Each structure type contains alternating [A2O2]2+ and Se2−/Se22− layers. Different ordered and disordered arrangements of Se2− and [Se–Se]2− give the Se layer flexibility and lead to the four different structure types observed. The volume coefficients of expansion for A4O4Se3 ranged from +1.746(9) × 10−5 to +2.237(3) × 10−5 K−1 from 12 to 300 K; no structural phase transitions were observed in this temperature range. Diffuse reflection spectra show A4O4Se3 are semiconductors with band gap Eg 1.02–1.46 eV. Gd4O4Se3, Dy4O4Se3, and Tb4O4Se3 samples show antiferromagnetic ordering with Néel temperature, TN, of 7–9 K. DFT calculations confirm the two different valence states of Se2− and Se22− in Eu4O4Se3.
- This article is part of the themed collection: In memory of Professor Kenneth Wade

 Please wait while we load your content...
Please wait while we load your content...