Quinoline-2-carboimine copper complex immobilized on amine functionalized silica coated magnetite nanoparticles: a novel and magnetically retrievable catalyst for the synthesis of carbamates via C–H activation of formamides†
Abstract
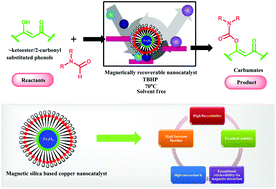
In the present study, we report the synthesis of a highly efficient and magnetically retrievable catalytic system (Cu-2QC@Am-SiO2@Fe3O4) through the covalent immobilization of quinoline-2-carboxaldehyde (2QC) on an amine functionalized silica coated ferrite nanosupport followed by metallation with copper acetate. The structure of the organic–inorganic hybrid nanomaterial has been confirmed using various physicochemical techniques such as Powder X-Ray Diffraction (XRD), Fourier Transform Infrared Spectroscopy (FT-IR), Scanning Electron Microscopy (SEM), Transmission Electron Microscopy (TEM), Energy Dispersive X-Ray Spectroscopy (EDS), Energy Dispersive X-Ray Fluorescence Spectroscopy (ED-XRF), Atomic Absorption Spectroscopy (AAS), Inductively Coupled Plasma Spectroscopy (ICP) and Vibrating Sample Magnetometry (VSM). The resulting nanocatalyst exhibits a remarkable catalytic efficacy in the synthesis of industrially and pharmaceutically significant carbamates via the C–H activation of formamides under solvent free conditions. The most important attribute of the present methodology is that the catalyst can be recovered simply through an external magnetic force and reused several times without any significant deterioration in its activity. Furthermore, the heterogeneity test has been carried out in order to ensure the intrinsic stability of the nanostructured catalyst. The activity of the Cu-2QC@Am-SiO2@Fe3O4 nanocatalyst has been found to be far more superior in comparison with the literature precedents in terms of the product yield, cost and reusability of the catalyst. Besides, ambient reaction conditions, simple workup procedure, wide substrate scope and cost effectiveness are some of the other outstanding features of this protocol that make it economical and sustainable.

 Please wait while we load your content...
Please wait while we load your content...