Exploring the decomposition pathways of iron asymmetric transfer hydrogenation catalysts†
Abstract
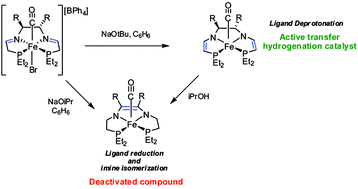
Our group has developed a series of iron-based asymmetric transfer hydrogenation (ATH) catalysts for the reduction of polar double bonds. The activation of the precatalysts as well as the catalytic mechanism have been thoroughly investigated, but the decomposition pathways of these systems are poorly understood. Herein, we report a study of the deactivation pathways for an iron ATH catalyst under catalytically relevant conditions. The decomposition pathways were examined using experimental techniques and density functional theory (DFT) calculations. The major decomposition products that formed, Fe(CO)((Et)2PCH2CH2CHCHNCH2CH2P(Et)2) (3a) and Fe(CO)((Et)2PCH2CH2C(Ph)C(Ph)NCH2CH2P(Et)2) (3b), had two amido donors as well as a C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond on the diamine backbone of the tetradentate ligand. These species were identified by NMR studies and one was isolated as a bimetallic complex with Ru(II)Cp*. Two minor iron hydride species also formed concurrently with 3a, as determined by NMR studies, one of which was isolated and contained a fully saturated ligand as well as a hydride ligand. None of the compounds that were isolated were found to be active ATH catalysts.
C bond on the diamine backbone of the tetradentate ligand. These species were identified by NMR studies and one was isolated as a bimetallic complex with Ru(II)Cp*. Two minor iron hydride species also formed concurrently with 3a, as determined by NMR studies, one of which was isolated and contained a fully saturated ligand as well as a hydride ligand. None of the compounds that were isolated were found to be active ATH catalysts.
- This article is part of the themed collection: Earth Abundant Element Compounds in Homogeneous Catalysis

 Please wait while we load your content...
Please wait while we load your content...