La1−xLnxH(O3PCH3)2 (Ln = Tb, Eu; 0 < x ≤ 1): an organic–inorganic hybrid with lanthanide chains and tunable luminescence properties†
Abstract
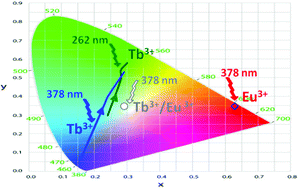
The organic/inorganic La1−xLnxH(O3PCH3)2 (Ln = Eu, Tb) hybrids have been synthesized by hydrothermal synthesis. The crystal structure of LaH(O3PCH3)2 consists of chains of edge-sharing LaO8 polyhedra linked through PO3C tetrahedra. Photoluminescence of Eu3+, Tb3+ and Eu3+/Tb3+ co-doped materials have been investigated. The Eu and Tb hybrids show no concentration quenching versus doping rate suggesting energy migration through a percolation model. The Eu hybrids exhibit a red emission while the Tb ones exhibit, with the Tb rate increasing, a blue to green emission under a 378 nm excitation wavelength and a cyan to green emission under a 262 nm excitation wavelength. The doping rate dependent red shift results from a cross relaxation phenomenon between closed Tb3+ ions. The blue to cyan shift observed for the slightly doped materials, when excitation wavelength shifts from 378 nm to 262 nm, is due to different relaxation phenomena, from the 5D3 level for a 378 nm excitation wavelength and from the 5D4 level via the 4f5d level for a 262 nm excitation wavelength. Under a 378 nm wavelength, the co-doped La0.93Eu0.03Tb0.04H(O3PCH3)2 hybrid exhibits a white/cyan emission with CIE coordinates equal to x = 0.29, y = 0.37.

 Please wait while we load your content...
Please wait while we load your content...