Coordination polymers of uranium(iv) terephthalates†
Abstract
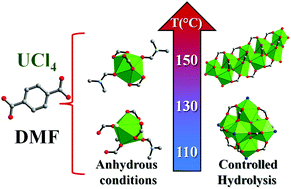
A series of tetravalent uranium terephthalates has been solvothermally synthesized in the solvent N,N-dimethylformamide (DMF) at temperature 100–150 °C with different water amounts. Composition diagrams have been determined for the U4+ metallic cation in the presence of terephthalic acid, and their crystal structures revealed the occurrence of two- or three-dimensional coordination polymers. In the absence of water, a mixture of two polytypes T-U2Cl2(bdc)3(DMF)4 (1) and M-U2Cl2(bdc)3(DMF)4 (2) has been identified at low temperature (100–110 °C) for bdc/U = 1–4 (bdc = terephthalate linker). Their structures are built up from isolated uranium centers in nine-fold coordination, surrounded by 6 carboxyl oxygen atoms, 2 oxygen atoms coming from DMF molecules and one chlorine atom. The uranium cations are linked to each other through the bdc ligand in order to generate a 3D framework. By increasing the temperature (130–150 °C), a layered like compound has been isolated, U2(bdc)4(DMF)4 (3). It is composed of discrete actinide centers in ten-fold coordination, with 8 carboxyl oxygen atoms and 2 oxygen atoms from DMF molecules. The connection of the UO10 units with the bdc linkers generates 2D sheets. When a controlled amount of water is added to the reaction medium, the crystallization of the UiO-66-like U6O4(OH)4(H2O)6(bdc)6·10DMF solid (containing a hexanuclear sub-unit) is observed for temperature 110–120 °C and the H2O/U molar ratio in the range of 2–10. At higher temperature (140–150 °C), a distinct phase appeared, U2O2(bdc)2(DMF) (4), which consists of infinite chains of uranium centers, linked to each other via the bdc ligands. Higher water contents led to the formation of urania UO2.
- This article is part of the themed collection: Dalton Discussion 14: Advancing the chemistry of the f-elements

 Please wait while we load your content...
Please wait while we load your content...