Mechanistic insight into the Z–E isomerization catalysis of azobenzenes mediated by bare and core–shell gold nanoparticles†
Abstract
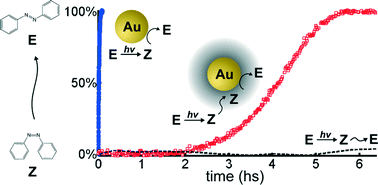
We explored the catalytic effect of 15 nm diameter gold nanoparticles (AuNPs) upon the thermal Z–E isomerization reaction of azobenzene and nine 4 and 4-4′ substituted azobenzenes (ABs). The kinetics follows a first order rate in ranges of [ABs] = 5 to 50 μM and [AuNPs] = 50 pM to 1 nM. A kinetic analysis of this compartmentalized system renders the thermal Z–E isomerization rate constant associated with each AuNP. Enhancements of 10- to 106-fold were measured for this rate constant in comparison to the same free ABs in solution. Experiments with selective Au facet coverage, as well as the kinetics studied in gold–silica core–shell nanoparticles (AuNP@SiO2) of different thicknesses, demonstrate the surface nature of the catalysis and allow one to evaluate the diffusion coefficient of azobenzene in the silica layer.

 Please wait while we load your content...
Please wait while we load your content...