Stable and selective electrochemical reduction of carbon dioxide to ethylene on copper mesocrystals†
Abstract
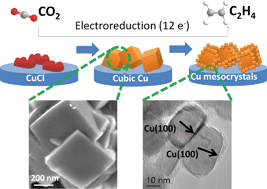
Stable and selective electrochemical reduction of carbon dioxide to ethylene was achieved using copper mesocrystal catalysts in 0.1 M KHCO3. The Cu mesocrystal catalysts were facilely derived by the in situ reduction of a thin CuCl film during the first 200 seconds of the CO2 electroreduction process. At −0.99 V vs. RHE, the Faradaic efficiency of ethylene formation using these Cu mesocrystals was ~18× larger than that of methane and forms up to 81% of the total carbonaceous products. Control CO2 reduction experiments show that this selectivity towards C2H4 formation could not be replicated by using regular copper nanoparticles formed by pulse electrodeposition. High resolution transmission electron microscopy reveals the presence of both (100)Cu facets and atomic steps in the Cu mesocrystals which we assign as active sites in catalyzing the reduction of CO2 to C2H4. CO adsorption measurements suggest that the remarkable C2H4 selectivity could be attributed to the greater propensity of CO adsorption on Cu mesocrystals than on other types of Cu surfaces. The Cu mesocrystals remained active and selective towards C2H4 formation for longer than six hours. This is an important and industrially relevant feature missing from many reported Cu-based CO2 reduction catalysts.

 Please wait while we load your content...
Please wait while we load your content...