Substitution of alcohols by N-nucleophiles via transition metal-catalyzed dehydrogenation
Abstract
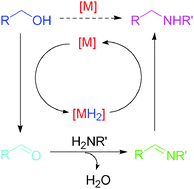
Transition metal-catalyzed substitution of alcohols by N-nucleophiles (or N-alkylation of amines and related compounds with alcohols) avoids the use of alkylating agents by means of borrowing hydrogen (BH) activation of the alcohol substrates. Water is produced as the only by-product, which makes the “BH” processes atom-economic and environmentally benign. Diverse types of homogeneous organometallic and heterogeneous transition metal catalysts, and substrates such as N-nucleophiles including amines, amides, sulfonamides and ammonia, and various alcohols, can be used for this purpose, demonstrating the promising potential of “BH” processes to replace the procedures using traditional alkylating agents in pharmaceutical and chemical industries. Borrowing hydrogen activation of alcohols for C–N bond formation has recently been paid more and more attention, and a lot of new and novel procedures and examples have been documented. This critical review summarizes the recent advances in “BH” substitution of alcohols by N-nucleophiles since 2009. “Semi-BH” N-alkylation processes with or without an external hydrogen acceptor are also briefly presented. Suitable discussion of the “BH” strategy provides new principles for establishing green processes to replace the relevant traditional synthetic methods for C–N bond formation.

 Please wait while we load your content...
Please wait while we load your content...