Uphill diffusion in multicomponent mixtures†
Abstract
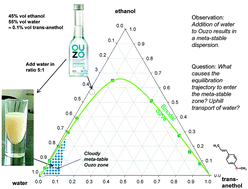
Molecular diffusion is an omnipresent phenomena that is important in a wide variety of contexts in chemical, physical, and biological processes. In the majority of cases, the diffusion process can be adequately described by Fick's law that postulates a linear relationship between the flux of any species and its own concentration gradient. Most commonly, a component diffuses down the concentration gradient. The major objective of this review is to highlight a very wide variety of situations that cause the uphill transport of one constituent in the mixture. Uphill diffusion may occur in multicomponent mixtures in which the diffusion flux of any species is strongly coupled to that of its partner species. Such coupling effects often arise from strong thermodynamic non-idealities. For a quantitative description we need to use chemical potential gradients as driving forces. The transport of ionic species in aqueous solutions is coupled with its partner ions because of the electro-neutrality constraints; such constraints may accelerate or decelerate a specific ion. When uphill diffusion occurs, we observe transient overshoots during equilibration; the equilibration process follows serpentine trajectories in composition space. For mixtures of liquids, alloys, ceramics and glasses the serpentine trajectories could cause entry into meta-stable composition zones; such entry could result in phenomena such as spinodal decomposition, spontaneous emulsification, and the Ouzo effect. For distillation of multicomponent mixtures that form azeotropes, uphill diffusion may allow crossing of distillation boundaries that are normally forbidden. For mixture separations with microporous adsorbents, uphill diffusion can cause supra-equilibrium loadings to be achieved during transient uptake within crystals; this allows the possibility of over-riding adsorption equilibrium for achieving difficult separations.

 Please wait while we load your content...
Please wait while we load your content...