Synthetic fluorescent probes for studying copper in biological systems
Abstract
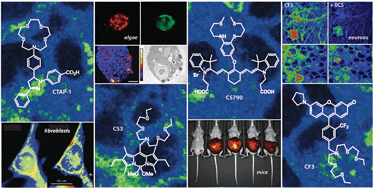
The potent redox activity of copper is required for sustaining life. Mismanagement of its cellular pools, however, can result in oxidative stress and damage connected to aging, neurodegenerative diseases, and metabolic disorders. Therefore, copper homeostasis is tightly regulated by cells and tissues. Whereas copper and other transition metal ions are commonly thought of as static cofactors buried within protein active sites, emerging data points to the presence of additional loosely bound, labile pools that can participate in dynamic signalling pathways. Against this backdrop, we review advances in sensing labile copper pools and understanding their functions using synthetic fluorescent indicators. Following brief introductions to cellular copper homeostasis and considerations in sensor design, we survey available fluorescent copper probes and evaluate their properties in the context of their utility as effective biological screening tools. We emphasize the need for combined chemical and biological evaluation of these reagents, as well as the value of complementing probe data with other techniques for characterizing the different pools of metal ions in biological systems. This holistic approach will maximize the exciting opportunities for these and related chemical technologies in the study and discovery of novel biology of metals.
- This article is part of the themed collection: Sensor Targets

 Please wait while we load your content...
Please wait while we load your content...