Electron transfer in peptides
Abstract
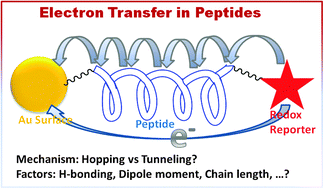
In this review, we discuss the factors that influence electron transfer in peptides. We summarize experimental results from solution and surface studies and highlight the ongoing debate on the mechanistic aspects of this fundamental reaction. Here, we provide a balanced approach that remains unbiased and does not favor one mechanistic view over another. Support for a putative hopping mechanism in which an electron transfers in a stepwise manner is contrasted with experimental results that support electron tunneling or even some form of ballistic transfer or a pathway transfer for an electron between donor and acceptor sites. In some cases, experimental evidence suggests that a change in the electron transfer mechanism occurs as a result of donor–acceptor separation. However, this common understanding of the switch between tunneling and hopping as a function of chain length is not sufficient for explaining electron transfer in peptides. Apart from chain length, several other factors such as the extent of the secondary structure, backbone conformation, dipole orientation, the presence of special amino acids, hydrogen bonding, and the dynamic properties of a peptide also influence the rate and mode of electron transfer in peptides. Electron transfer plays a key role in physical, chemical and biological systems, so its control is a fundamental task in bioelectrochemical systems, the design of peptide based sensors and molecular junctions. Therefore, this topic is at the heart of a number of biological and technological processes and thus remains of vital interest.
- This article is part of the themed collection: Molecular Wires

 Please wait while we load your content...
Please wait while we load your content...