Incorporating single molecules into electrical circuits. The role of the chemical anchoring group
Abstract
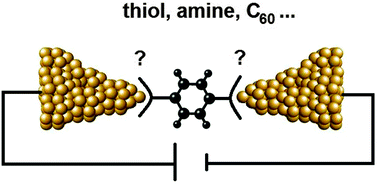
Constructing electronic circuits containing singly wired molecules is at the frontier of electrical device miniaturisation. When a molecule is wired between a pair of electrodes, the two points of contact are determined by the chemical anchoring groups, located at the ends of the molecule. At this point, when a bias is applied, electrons are channelled from a metallic environment through an extremely narrow constriction, essentially a single atom, into the molecule. The fact that this is such an abrupt change in the electron pathway makes the nature of the chemical anchoring groups critically important regarding the propagation of electrons from the electrode across the molecule. A delicate interplay of phenomena can occur when a molecule binds to the electrodes, which can produce profound differences in conductance properties depending on the anchoring group. This makes answering the question “what is the best anchoring group for single molecule studies” far from straight forward. In this review, we firstly take a look at techniques developed to ‘wire-up’ single molecules, as understanding their limitations is key when assessing a molecular wire's performance. We then analyse the various chemical anchoring groups, and discuss their merits and disadvantages. Finally we discuss some theoretical concepts of molecular junctions to understand how transport is affected by the nature of the chemical anchor group.
- This article is part of the themed collection: Molecular Wires

 Please wait while we load your content...
Please wait while we load your content...