Ligand influence on the electronic spectra of monocationic copper–bipyridine complexes†
Abstract
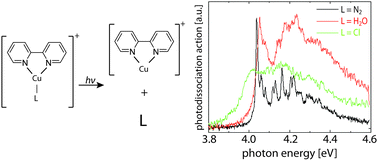
We present photodissociation spectroscopy and computational analysis of three monocationic Cu–bipyridine complexes with one additional ligand of different interaction strength (N2, H2O and Cl) in the visible and UV. All three complexes show similar ππ* bands with origins slightly above 4 eV and vibrational band contours that are due to bipyridine ring deformation modes. Experiments at low temperature show that excited-state lifetime is the limiting factor for the width of the vibrational features. In the case of Cl as a ligand, there is a lower lying bright ligand-to-ligand charge-transfer state around 2.75 eV. The assignment of the transitions was made based on equation-of-motion coupled-cluster calculations. While the nature of the ligand does not significantly change the position of the bright ππ* state, it drastically changes the excited-state dynamics.

 Please wait while we load your content...
Please wait while we load your content...