Increase in the photoreactivity of uracil derivatives by doubling thionation†
Abstract
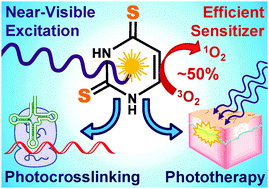
The ability of 4-thiouracil to strongly absorb UVA radiation and to populate a reactive triplet state in high yield has enabled its use as a versatile photocrosslinker for nearly 50 years. In this contribution, we present a detailed spectroscopic and photochemical investigation of the 2-thiouracil, 4-thiouracil, and 2,4-dithiouracil series in an effort to further advance this chemistry and to scrutinize the photoreactivity of 2,4-dithiouracil. Our results reveal that excitation of 2,4-dithiouracil leads to intersystem crossing to the triplet manifold in 220 ± 40 fs, which enables the population of the reactive triplet state with near unity yield (ΦT = 0.90 ± 0.15) and ultimately leads to a ca. 50% singlet oxygen generation (ΦΔ = 0.49 ± 0.02)—one of the highest singlet oxygen yields reported to date for a photoexcited thiobase. In addition, the long-lived triplet state of 2,4-dithiouracil reacts efficiently with the nucleic acid base adenine 5′-monophosphate through a direct, oxygen-independent photocycloaddition mechanism and at a rate that is at least 3-fold faster than that of 4-thiouracil under equal conditions. The new physico-chemical insights reported for these RNA-thiobase derivatives are compared to those of the DNA and RNA bases and the DNA-thiobase derivatives. Furthermore, the strong near-visible absorption and increased photoreactivity measured for 2,4-dithiouracil lays a solid foundation for developing RNA-targeted photocrosslinking and phototherapeutic agents that are more effective than those currently available.

 Please wait while we load your content...
Please wait while we load your content...