Anion exchange in ionic liquid mixtures
Abstract
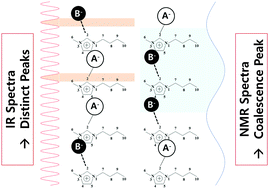
Advantage of ionic liquids as designer solvents can be maximized by mixing different ionic liquids (ILs) for a possibility of continuous tunability of material properties. The property of these mixed ILs would be determined by their microscopic conformation between cations and anions. The mixtures of two ILs having 1-butyl-3-methylimidaolium cations and different anions were investigated by IR and NMR spectroscopy, utilizing that the vibrational frequencies of the C–H stretching and bending modes of the most acidic proton in the imidazolium ring of the cation and the NMR chemical shift for the corresponding proton were clearly distinct between the ILs having different anions. The IR absorption spectra of the IL mixtures at different concentrations were well-matched to weighted sums of the two spectra of the pure ILs. In contrast, the two distinct peaks in the NMR spectra of the pure ILs coalesced into a single peak, which shifted continuously following the relative portion of two different ILs in the mixture. IR spectroscopy in the optical frequency range seems to take the instantaneous snapshot of the cation–anion interaction, while NMR spectroscopy in the radio-frequency (∼500 MHz) range samples over a much longer timescale, enough for the cation to interact with different anion species in the mixture.

 Please wait while we load your content...
Please wait while we load your content...