Photoswitching the mechanical properties in Langmuir layers of semifluorinated alkyl-azobenzenes at the air–water interface†
Abstract
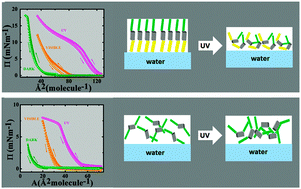
Semifluorinated alkyl-azobenzene derivatives (SFAB) can form stable Langmuir layers at the air–water interface. These systems combine the amphiphobic character of semifluorinated alkyl units as structure-directing motifs with photochromic behavior based on the well-known reversible cis–trans isomerization upon irradiation with UV and visible light. Herein, we report our investigations of the structural and dynamic tunability of these SFAB layers at the air–water interface in response to an external light stimulus. The monolayer structures and properties of [4-(heptadecafluorooctyl)phenyl](4-octylphenyl)diazene (F8-azo-H8) and bis(4-octylphenyl)diazene (H8-azo-H8) were studied by neutron reflectivity, surface pressure–area isotherms with compression–expansion cycles, and interfacial rheology. We find that UV irradiation reversibly influences the packing behavior of the azobenzene molecules and interpret this as a transition from organized layer structures with the main axis of the molecule vertically oriented in the trans form to random packing of the cis isomer. Interestingly, this trans–cis isomerization leads to an increase in surface pressure, which is accompanied by a decrease in viscoelastic moduli. These results suggest ways of tailoring the properties of responsive fluid interfaces.


 Please wait while we load your content...
Please wait while we load your content...