Reaction kinetics for the solid state synthesis of the AlH3/MgCl2 nano-composite by mechanical milling†
Abstract
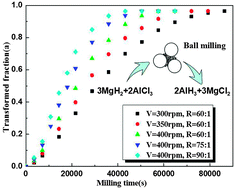
The process of mechanical milling has been proved to be a cost-effective way to synthesize the AlH3/MgCl2 nano-composite by using MgH2 and AlCl3 as reagents. However, so far there is no comprehensive knowledge of the kinetics of this process. In an effort to predict the reaction progress and optimize the milling parameters, the kinetics of the synthesis of the AlH3/MgCl2 nano-composite by mechanical milling of MgH2 and AlCl3 is experimentally investigated in the present work. The reaction progress or the transformation fraction upon milling for different times is evaluated using the isothermal hydrogen desorption test of the as-milled samples at 220 °C, which is much lower than the threshold temperature for the de-hydriding of the reagent MgH2 but enough for the de-hydriding of the as-synthesized nano-sized AlH3. The effects of milling parameters on the reaction kinetics as well as the underlying mechanism are discussed by referring to the mechanical energy input intensity, the vial temperature and the Gibbs free energy change for the reaction. Furthermore, it is found that the Johnson–Mehl–Avrami (JMA) model can well describe the kinetics theoretically. By fitting the experimental data with the JMA expression, the theoretical kinetics expressions, the equation parameters, and the activation energy are obtained.

 Please wait while we load your content...
Please wait while we load your content...