The effect of oleic acid on the synthesis of Fe3−xO4 nanoparticles over a wide size range†
Abstract
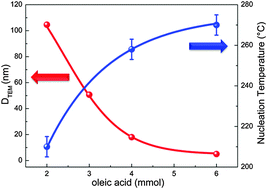
This work reports on the effect of the oleic acid concentration on the magnetic and structural properties of Fe3−xO4 nanoparticles synthesized by thermal decomposition of Fe(acac)3 in benzyl-ether. This method allows the synthesis of highly monodisperse particles ranging from 7 to 100 nm in size by only varying the concentration of oleic acid in the reaction mixture. The structural and magnetic characterization reveal homogeneous particles in composition, with narrow particle size distribution, which are single-phase magnetite with almost bulk-like values of the saturation magnetization of about 90–99 emu g−1 at low temperatures and show the characteristic anomaly in the zero field-cooling magnetization curves associated with the Verwey transition for nanoparticles bigger than about 7 nm. In addition, the analyses of aliquots of the reaction mixtures by Fourier transform infrared spectroscopy at various stages shed light on the nucleation and growth processes of the particles.

 Please wait while we load your content...
Please wait while we load your content...