Structure determination of trans-cinnamaldehyde by broadband microwave spectroscopy†
Abstract
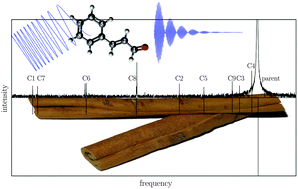
The rotational spectrum of trans-cinnamaldehyde ((E)-3-phenyl-2-propenal, C9H8O) was recorded by chirped-pulse Fourier transform microwave spectroscopy in the frequency range of 2–8.5 GHz. The odourant molecule is the essential component of cinnamon oil and causes the characteristic smell. The rotational signatures of two conformers were observed: s-trans–trans- and s-cis–trans-cinnamaldehyde. The rotational spectra of s-trans–trans-cinnamaldehyde and all of its 13C-monosubstituted species in natural abundance were assigned and the corresponding carbon backbone structure was determined. The second conformer s-cis–trans-cinnamaldehyde is about 9 kJ mol−1 higher in energy and could also be identified in the spectrum.

 Please wait while we load your content...
Please wait while we load your content...