Cu(i) stabilizing crosslinked polyethyleneimine
Abstract
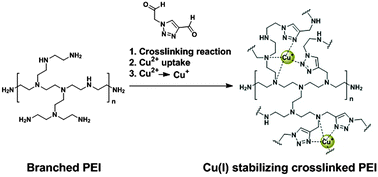
Polyethyleneimine (PEI) is known for its metal-coordinating properties and in its crosslinked form has applications in different areas ranging from drug delivery to waste water treatment and recovery of trace metals. With the aim to regulate the coordination environment of Cu(I) and Cu(II) ions in marine coatings, we have prepared a triazole cross-linking agent with ‘soft’ coordination that can crosslink PEI via indirect reductive amination. We have shown that this triazole crosslinker not only increases the Cu(II) absorption capacity of the crosslinked PEI in comparison to the traditionally used glutaraldehyde-crosslinked PEI (PEI-GA), but also allows the crosslinked polymer network to stabilize the Cu(I) oxidation state more effectively. The Cu(II) uptake and Cu(I) stabilization of the polymer have been determined by elemental analysis and UV-vis spectroscopy. It was found that the triazole-crosslinked polymer (PEI-TA) could coordinate up to 12 wt% of Cu(II) before and 6 wt% Cu(II) after imine reduction.

 Please wait while we load your content...
Please wait while we load your content...