Resolving the ambiguity in the relation between Stokes shift and Huang–Rhys parameter
Abstract
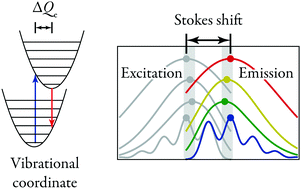
Electronic transitions in luminescent molecules or centers in crystals couple to vibrations. This results in broadening of absorption and emission bands, as well as in the occurence of a Stokes shift EStokes. In principle, one can derive from EStokes the Huang–Rhys parameter S, which describes the microscopic details of the vibrational coupling and can be related to the equilibrium position offset ΔQe between the ground state and excited state. The commonly used textbook relations EStokes = (2S − 1)ℏω and EStokes = 2Sℏω are only approximately valid. In this paper we investigate how EStokes is related to S, taking into account the effects of a finite temperature. We show that in different ranges of temperature, different approximate relations between EStokes and S are appropriate. Moreover, we demonstrate that the difference between the barycenters of absorption and emission bands can be used to determine S in an unambiguous way. The position of the barycenter is, contrary to the Stokes shift, unaffected by temperature.

 Please wait while we load your content...
Please wait while we load your content...