Ion specific effects: decoupling ion–ion and ion–water interactions
Abstract
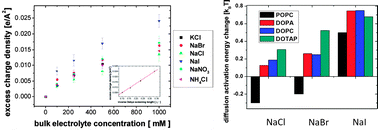
Ion-specific effects in aqueous solution, known as the Hofmeister effect, are prevalent in diverse systems ranging from pure ionic to complex protein solutions. The objective of this paper is to explicitly demonstrate how complex ion–ion and ion–water interactions manifest themselves in the Hofmeister effect based on a series of recent experimental observations. These effects are not considered in the classical descriptions of ion effects, such as the Derjaguin–Landau–Verwey–Overbeek (DLVO) theory, and therefore they fail to describe the origin of the phenomenological Hofmeister effect. However, given that models considering the basic forces of electrostatic and van der Waals interactions can offer rationalization for the core experimental observations, a universal interaction model stands a chance of being developed. In this perspective, we separately derive the contribution from ion–ion electrostatic interactions and ion–water interactions from second harmonic generation (SHG) data at the air–ion solution interface, which yields an estimate of the ion–water interactions in solution. The Hofmeister ion effect observed for biological solutes in solution should be similarly influenced by contributions from ion–ion and ion–water interactions, where the same ion–water interaction parameters derived from SHG data at the air–ion solution interface could be applicable. A key experimental data set available from solution systems to probe ion–water interactions is the modulation of water diffusion dynamics near ions in a bulk ion solution, as well as near biological liposome surfaces. This is obtained from Overhauser dynamic nuclear polarization (ODNP), a nuclear magnetic resonance (NMR) relaxometry technique. The surface water diffusivity is influenced by the contribution from ion–water interactions, both from localized surface charges and adsorbed ions, although the relative contribution of the former is larger on liposome surfaces. In this perspective, ion–water interaction energy values derived from experimental data for various ions are compared with theoretical values in the literature. Ultimately, quantifying ion-induced changes in the surface energy for the purpose of developing valid theoretical models for ion–water interactions will be critical to rationalizing the Hofmeister effect.
- This article is part of the themed collection: Bunsentagung 2015: Solvation Science

 Please wait while we load your content...
Please wait while we load your content...