Electro-catalytic oxidation of hemicelluloses at the Au electrode†
Abstract
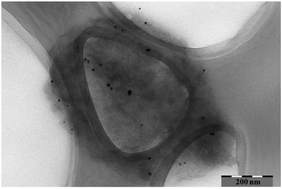
The recently reported electro-catalytic reaction mechanism for the oxidation of cellulose is proposed to work also for the electro-oxidation of other polysaccharides (e.g. hemicelluloses). In this report, the electrochemical reactivity of some hemicelluloses (xylan, arabinoxylan, glucomannan, xyloglucan, and glucuronoxylan) in 1.3 M NaOH solution is described. The electrochemical property of each of the studied hemicelluloses at the Au electrode surface was characterized by cyclic voltammetry. Xylan, xyloglucan and glucuronoxylan were found to be electrochemically active. Electrochemical properties of xylan were further studied by electrochemical impedance spectroscopy. The electrochemical reaction products of xylan, obtained by extensive electrolysis of the xylan solution by cyclic voltammetry, were characterized by FTIR spectroscopy, 13C-NMR spectroscopy, SEM and TEM. The structural studies suggest that the oxidized xylan is a functional material where some of the OH-groups have been oxidized to carboxyl groups making that part of the oxidation product soluble in water under ambient conditions (23 °C, pH 7). The other part remains insoluble in water and contains Au nanoparticles. This work indicates that other polysaccharides than cellulose can also be electrochemically oxidized.

 Please wait while we load your content...
Please wait while we load your content...