The isotope method for the determination of stoichiometry between compounds forming the polypyrrole and glucose oxidase composite
Abstract
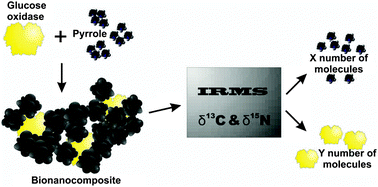
Stable isotope ratio mass spectrometry is a conventional method used in archaeology, and medical, environmental and paleoenvironmental reconstruction studies. However new insights and applicability of the equipment often open new research areas and improve our understanding of the ongoing processes. Therefore the stable isotope ratio mass spectrometry method was applied for the stoichiometry determination of the complex polypyrrole and glucose oxidase composite (PPy–GOx composite). The enzyme glucose oxidase and conducting polymer polypyrrole were reported to form a composite, which was evaluated in time using the dynamic light scattering method. The consistent enlargement of the PPy–GOx composite and the relative decrease of the spare enzyme molecules were observed in the polymerization solution. UV-VIS spectrometry was employed to follow the polymerization process. The isotope mixing model was applied for the evaluation of the constitution of the PPy–GOx composite. According to the obtained results the determination of the PPy–GOx composite stoichiometry could be more reliably determined using the nitrogen isotope ratio approach in comparison to the carbon approach. We expect that this novel work will widen the applications of stable isotope ratio mass spectrometry in research.

 Please wait while we load your content...
Please wait while we load your content...