Photoresponsive ionic liquid crystals based on azobenzene guanidinium salts†
Abstract
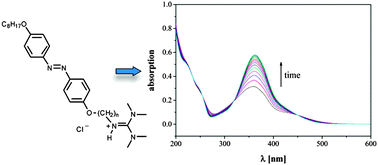
The use of non-ionic LC phases as anisotropic matrices for E/Z-isomerization of azo-guest molecules is often restricted due to limited solubilities and demixing effects. In this study we therefore employed an ionic liquid crystal (ILC) matrix to follow the photo-induced E/Z-isomerization of ionic mesogenic azobenzene guanidinium guests. The latter were prepared from 4-hydroxy-4′-(octyloxy)azobenzene, which was first treated with N-(bromoalkyl)phthalimides to introduce the spacer with varying chain length. Removal of phthalimide and final reaction with a formamidinium salt linked the ionic head group to the photoisomerizable azobenzene unit. Investigation of the mesomorphic behaviour revealed for all azobenzene ILCs smectic A mesophases with high translational order parameters and partial bilayers, as could be stated by layer spacing d. Similar packing behaviour was found for the solid state by X-ray crystal structure analysis. E/Z-isomerization of azobenzene ILCs which were completely miscible with the ionic LC phase of C12MIM-Br as anisotropic host was induced by irradiation with UV light and the reisomerization observed by time-resolved UV-Vis spectroscopy. For comparison, water was used as isotropic host. Z/E-reisomerization activation energies exhibited similar values of 97–100 kJ mol−1 irrespective of spacer lengths and the type of host. The results demonstrate that a proper match of steric requirements of host and guest as well as layer spacings are needed for a decreased activation energy.
- This article is part of the themed collection: Bunsentagung 2015: Solvation Science

 Please wait while we load your content...
Please wait while we load your content...