Water-induced phase transformation of a CuII coordination framework with pyridine-2,5-dicarboxylate and di-2-pyridyl ketone: synchrotron radiation analysis†
Abstract
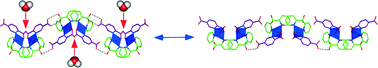
Phase transformations in solid coordination frameworks (SCFs) are of interest for several applications, and this work reports a crystal-to-crystal transformation found for hydrogen-bonded CuII-based solid coordination frameworks (SCFs). Thus, the combination of PDC and (py)2C(OH)2 ligands, where PDC is pyridine-2,5-dicarboxylate and (py)2C(OH)2 is the derivative gem-diol of di-2-pyridyl ketone ((py)2CO), produces [Cu(PDC)((py)2C(OH)2)(H2O)] (1). Compound 1 transforms into [Cu(PDC)((py)2C(OH)2)] (2) by thermally-induced dehydration. Characterization of both compounds has been carried out by means of IR spectroscopy, single crystal and powdered sample X-ray diffraction (XRD) through conventional and synchrotron radiation, thermogravimetry (TG), X-ray thermodiffractometry (TDX), and scanning electron microscopy (SEM). Since the molecules of water in 1 are coordinated to the metal ions, their removal induces local distortions on the coordination sphere (square pyramidal for 1 and square planar for 2), which extend through the whole framework, affecting the hydrogen bond system and the packing. In fact, the wavy nature of the planes in 1 becomes sharper in 2, producing an oscillation of the framework, i.e., from open (1) to close (2) waves. The crystal-to-crystal transformation is reversible (1 ↔ 2) and hysteresis has been observed associated with it. Quantum-mechanical calculations based on density functional theory (DFT) show that the 1 ↔ 2 structural rearrangement involves a high amount of energy, which means that the role of the coordinated molecule of water exceeds the mere formation of hydrogen bonds.

 Please wait while we load your content...
Please wait while we load your content...