Single-step synthesis and catalytic activity of structure-controlled nickel sulfide nanoparticles
Abstract
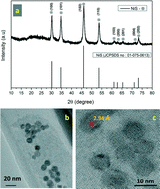
Nanoparticle single-phase nickel sulfides such as NiS, NiS2, Ni3S4, and Ni7S6 were prepared from elemental sulfur and nickel nitrate hexahydrate, using a temperature-controlled precursor injection method. The initial ratio of the concentrations of the sources was used to control the size and phase of the final product. Phase control was confirmed using X-ray diffraction and transmission electron microscopy. The synthesized nickel sulfide phases, which had metallic characteristics, were used to study the catalytic reduction of 4-nitrophenol. The results showed that the catalytic activity of the NiS nanoparticles in the reduction of 4-nitrophenol to 4-aminophenol was higher than those of the other nickel sulfide phases. In addition, the nanocrystals showed good separation ability and reusability for reduction of 4-nitrophenol.

 Please wait while we load your content...
Please wait while we load your content...