Hydrate smaller than the anhydrate†
Abstract
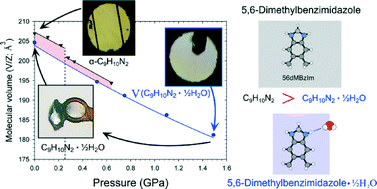
Anhydrous 5,6-dimethylbenzimidazole (dMBzIm) crystals are obtained from an aqueous solution at ambient pressure, but above 0.25 GPa the hemihydrate dMBzIm·½H2O is formed. It can be stored for over a year in open vials. Counter-intuitively, the volume of dMBzIm·½H2O is smaller than that of the anhydrate. Ambient and high-pressure structures of dMBzIm and dMBzIm·½H2O crystals grown in situ in a diamond-anvil cell have been determined by X-ray diffraction methods. The isostructural phase transition observed in dMBzIm at 0.25 GPa coincides with the changed preference to hemihydrate crystallization. The significantly different ambient and high-pressure hydration preferences of imidazole and benzimidazole derivatives have been explained by the interplay of interactions and the efficiency of crystal packing measured by the size and volume of voids.

 Please wait while we load your content...
Please wait while we load your content...