In situ IR spectral identification of NH4H2PO4 structural evolution during crystallization in water–ethanol mixed solvent†
Abstract
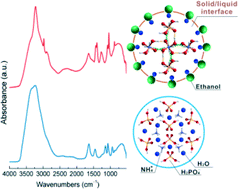
Both the crystallizing molecule without long-range ordered arrangement and the complex interactions between solvent, anti-solvent, and molecule crystal challenge the imaging of molecule configurations during anti-solvent crystallization. Here, the structural evolution of NH4H2PO4 during anti-solvent crystallization was tracked by in situ ATR-IR spectroscopy. The NH4H2PO4 molecule undergoes D2d → C2v → D2d or C2v → D2d during anti-solvent crystallization, depending on both V(H2O)/V(ethanol) ratio and the concentration of NH4H2PO4 aqueous solution. Moreover, IR spectroscopy can also be used to confirm the anti-solvent role of ethanol in the NH4H2PO4–H2O–ethanol crystallization system. The volatilization of ethanol has been demonstrated as an effective approach to control the dissolution process of NH4H2PO4, which can be used to study the crystallization–dissolution–recrystallization process. The present study can shed light on the anti-solvent crystallization physical and chemistry of NH4H2PO4 from a molecule-level viewpoint.

 Please wait while we load your content...
Please wait while we load your content...