Isolation and crystal structure of a dithiophene dication: controlling covalent connection and disconnection with temperature and phase†
Abstract
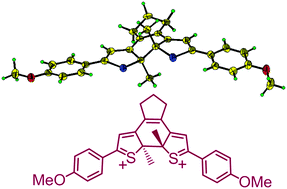
A dithienylethene (1o) undergoes a two-electron chemical oxidation to a singlet diradical as an open-isomer (1o2+) in solution, which cyclizes to a closed-form (1c2+) upon cooling. The latter crystallizes out and its structure is analyzed by single crystal X-ray diffraction. Equilibrium between 1o2+ and 1c2+ in solution is observed by NMR and UV spectroscopy at various temperatures and is further supported by reduction reactions with Zn powder.

 Please wait while we load your content...
Please wait while we load your content...