Two-dimensional bricklayer arrangements of tolans using halogen bonding interactions†
Abstract
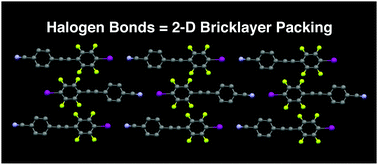
Diphenylacetylene (tolan) derivatives with self-complementary aryl halides and halogen bond-accepting nitriles form 2D bricklayer packing motifs when halogen bonding occurs. When halogen bonding is absent, as occurred with fluorinated aryl bromides, the molecules adopt other packing motifs. These results suggest halogen bonding is potentially useful for producing rarely observed 2D bricklayer motifs in organic semiconductors.

 Please wait while we load your content...
Please wait while we load your content...