Rational design of a photo-crosslinking BODIPY for in situ protein labeling†
Abstract
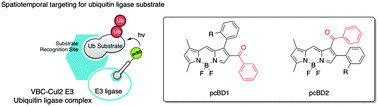
Photo-crosslinking agents have emerged as critical tools to investigate protein–protein interactions in complex proteomes, but there are few photocrosslinkers available at the moment. Here, we report the first rational design of a photo-crosslinking BODIPY fluorophore (pcBD) and its biological application for biomolecule labeling. As a photosensitizing functional motif, an aryl ketone group was incorporated into the BODIPY fluorophore, and a series of proteins were labeled by pcBD compounds upon UV irradiation. In order to investigate protein–protein interactions in a protein mixture, amino-functionalized pcBD was prepared and covalently attached to a ubiquitin ligase binding peptide. Upon UV irradiation, we could successfully visualize the substrates in the total lysate. These results provided a proof of concept for spatially controllable tagging via photo-activation of the pcBD scaffold, and demonstrated its potential usage for in situ labeling applications.


 Please wait while we load your content...
Please wait while we load your content...