An ultrasensitive and selective turn-off fluorescent nanoprobe for the detection of copper ions†
Abstract
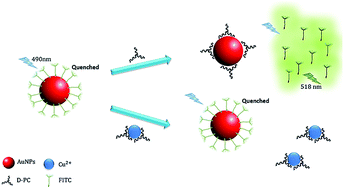
In this study, a novel approach for sensitive and extremely selective detection of copper ions has been developed based on fluorescence resonance energy transfer (FRET) between AuNPs as an acceptor and fluorescein isothiocyanate (FITC) as a donor. Initially, the fluorescence of the FITC molecule turns-off due to adsorption to the AuNPs surface. However, the fluorescence of the FITC–AuNPs system was switched to turn-on by adding D-Pencillamine (D-PC) to the solution mixtures as FITC molecules are released upon displacement by D-PC on the NPs surface. The higher affinity of D-PC toward the surface of AuNPs was further evidenced by controlling the fluorescence intensity enhancement of the released FITC in the presence of copper ions. In this scenario, the chelating agent of the D-PC strongly interacts with copper ions, thereby becoming less competitive for adsorption to the AuNPs surface. This methodology was implemented for the determination of copper ions, where under the optimum experimental conditions (pH 8, [FITC] = 1.0 μM, [D-PC] = 6.25 μM), two linear calibration curves were obtained within the range of 1–9 nM and 10–40 nM with a detection limit of 0.3 nM. Ultimately, the designed nanoprobe was successfully employed for the detection of copper ions in food and water samples.

 Please wait while we load your content...
Please wait while we load your content...