Microemulsion electrokinetic chromatography with polarity switching stacking mode for the determination of dexamethasone and dexamethasone sodium phosphate: application to pharmacokinetic studies in rabbit plasma
Abstract
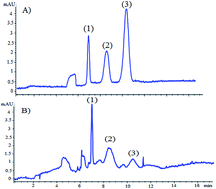
A novel method based on microemulsion electrokinetic chromatography (MEEKC) with reversed electrode polarity stacking mode (REPSM) was developed and validated for the simultaneous determination of dexamethasone sodium phosphate (DEX-SP) and the active drug dexamethasone (DEX) in rabbit plasma. The optimum separation was achieved when a microemulsion background electrolyte consisting of 30 mM borate buffer (pH 9.2), 20 mM sodium dodecyl sulfate (SDS), 0.8% ethyl acetate and 2% 1-butanol was used. Electrophoretic separation was carried out at 24 °C and at +23 kV. A stacking technique has been developed to pre-concentrate samples and to increase the amount of sample that can be loaded onto the capillary without degrading the separation, up to 160 s. REPSM was applied using the following optimized program: +23 kV (0–0.6 min) then −23 kV (0.6–1.5 min) and finally +23 kV for the rest of the run. The analytes were base-line resolved within 12 min. Using hydrochlorothiazide (HCT) as the internal standard, the peak area ratios were found to be linear in the concentration range of 0.1–2.0 μg mL−1 for DEX-SP and 0.05–2.0 μg mL−1 for DEX. The applied stacking mode increased the detectability of the method by about 40-fold with limits of detection and quantitation of 0.03 and 0.1 μg mL−1 for DEX-SP, and 0.02 and 0.05 μg mL−1 for DEX, respectively. The developed method was fully validated according to the ICH guidelines. This optimized method was applied for the determination of DEX-SP and DEX in rabbit plasma samples previously treated with I.V. injection of DEX-SP. The in vivo results thus obtained were used to study the pharmacokinetics of the cited drugs in rabbit plasma.

 Please wait while we load your content...
Please wait while we load your content...