Selective determination of mefenamic acid in the presence of 1000-fold excess paracetamol and caffeine using a multiwalled carbon nanotube–polymer composite electrode†
Abstract
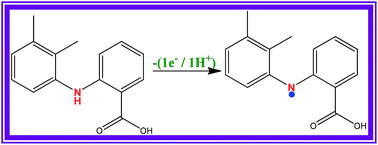
This article describes the selective and sensitive determination of mefenamic acid (MA) using a carboxylated multiwalled carbon nanotube (FMWCNT)-nanostructured conducting polymer (p-ATT) composite modified glassy carbon (GC) electrode in 0.2 M phosphate buffer solution (PBS, pH 7.2). The bare GC electrode failed to show a stable response to MA oxidation due to the surface fouling caused by the oxidized product of MA. However, the FMWCNT/p-ATT composite electrode showed a 2.4-fold higher oxidation current with 70 mV less positive potential for MA when compared to the bare GC electrode. The higher electrocatalytic activity of MA at the composite modified electrode may be due to the hydrogen bonding and electrostatic interactions between p-ATT and MA besides the π–π interaction between the FMWCNT and p-ATT. Further, the FMWCNT/p-ATT composite electrode showed extreme selectivity towards MA in the presence of 1000-fold excess of paracetamol (PA) and caffeine (CAF) and 3330-fold excess of other common interferents. The amperometric current response was increased linearly with increasing MA concentration in the range of 40–5000 nM with a correlation coefficient of 0.9980 and the limit of detection was found to be 90 pM (S/N = 3). The practical application of the present modified electrode was successfully demonstrated by determining MA in commercial drug samples.

 Please wait while we load your content...
Please wait while we load your content...