Continuous flow microfluidic solution for quantitative analysis of active pharmaceutical ingredient content and kinetic release†
Abstract
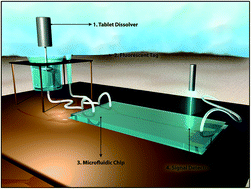
Counterfeit and substandard medicines are a grave public health concern that comprises a multibillion-dollar black market and claims over 100 000 lives every year. The World Health Organization estimates that 1–50% of medicines in countries around the world are adulterated, and their presence imposes serious financial and economic burdens while also contributing to the rise of drug-resistant pathogens. Although there are a number of technologies available for field-based quality screening, none can reliably quantify active pharmaceutical ingredient (API) content or kinetic release from a dissolving tablet at the point of care. This work presents an alternative analytical technique to address this major gap. Our system can specifically and accurately quantify drug API content and kinetic release using a portable, inexpensive, and easy-to-use aptamer based fluorescence platform. We demonstrate that aptamers can provide a simple and effective way to target a wide range of APIs, while maintaining high quantitative precision and accuracy. A microfluidic, flow-through system is employed to obtain drug quality information using a single step procedure that shows an accuracy of over 97% for both API quantification and kinetic release.
- This article is part of the themed collection: Microfluidics Research 2015-2016

 Please wait while we load your content...
Please wait while we load your content...