Evaluation of separation properties of a modified strong cation exchange material named MEX and its application in 2D-MEX × C18 system to separate peptides from scorpion venom
Abstract
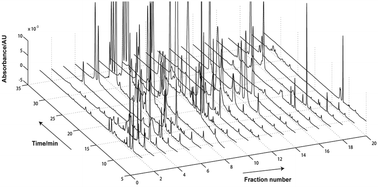
Peptides from scorpion venom represent one of the most promising drug sources for drug discovery for some specific diseases. Current challenges in their separation include high complexity, high homologies and the huge range of peptides. In this paper, a modified strong cation exchange material, named MEX, was utilised for the two-dimensional separation of peptides from complex scorpion venom. The silica-based MEX column was bonded with two functional groups; benzenesulfonic acid and cyanopropyl. To better understand its separation mechanisms, seven standard peptides with different properties were employed in an evaluation study, the results of which showed that two interactions were involved in the MEX column: electrostatic interactions based on benzenesulfonic acid groups dominated the separation of peptides; weak hydrophobic interactions introduced by cyanopropyl groups increased the column's selectivity for peptides with the same charge. This characteristic allowed the MEX column to overcome some of the drawbacks of traditional strong cation exchange (SCX) columns. Furthermore, the study showed the great effects of the acetonitrile (ACN) content, the sodium perchlorate (NaClO4) concentration and the buffer pH in the mobile phase on the peptides’ retention and separation selectivity on the MEX column. Subsequently, the MEX column was combined with a C18 column to establish an off-line 2D-MEX × C18 system to separate peptides from scorpion Buthus martensi Karsch (BmK) venom. Due to complementary separation mechanisms in each dimension, a high orthogonality of 47.62% was achieved. Moreover, a good loading capacity, excellent stability and repeatability were exhibited by the MEX column, which are beneficial for its use in future preparation experiments. Therefore, the MEX column could be an alternative to the traditional SCX columns for the separation of peptides from scorpion venom.

 Please wait while we load your content...
Please wait while we load your content...