Raman micro spectroscopy for in vitro drug screening: subcellular localisation and interactions of doxorubicin
Abstract
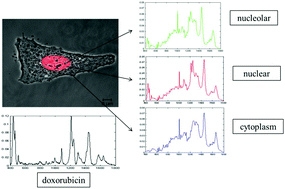
Vibrational spectroscopy, including Raman micro spectroscopy, has been widely used over the last few years to explore potential biomedical applications. Indeed, Raman micro spectroscopy has been demonstrated to be a powerful non-invasive tool in cancer diagnosis and monitoring. In confocal microscopic mode, the technique is also a molecularly specific analytical tool with optical resolution which has potential applications in subcellular analysis of biochemical processes, and therefore as an in vitro screening tool of the efficacy and mode of action of, for example, chemotherapeutic agents. In order to demonstrate and explore the potential in this field, established, model chemotherapeutic agents can be valuable. In this study paper, Raman micro spectroscopy coupled with confocal microscopy were used for the localization and tracking of the commercially available drug, doxorubicin (DOX), in the intracellular environment of the lung cancer cell line, A549. Cytotoxicity assays were employed to establish clinically relevant drug doses for 24 h exposure, and Confocal Laser Scanning Fluorescence Microscopy was conducted in parallel with Raman micro spectroscopy profiling to confirm the drug internalisation and localisation. Multivariate statistical analysis, consisting of PCA (principal components analysis) was used to highlight doxorubicin interaction with cancer cells and spectral variations due to its effects before and after DOX spectral features subtraction from nuclear and nucleolar spectra, were compared to non-exposed control spectra. Results show that Raman micro spectroscopy is not only able to detect doxorubicin inside cells and profile its specific subcellular localisation, but, it is also capable of elucidating the local biomolecular changes elicited by the drug, differentiating the responses in different sub cellular regions. Further analysis clearly demonstrates the early apoptotic effect in the nuclear regions and the initial responses of cells to this death process, demonstrating the potential of the technique to monitor the mechanisms of action and response on a molecular level, with subcellular resolution.

 Please wait while we load your content...
Please wait while we load your content...