Synergistic interaction between lipid-loading and doxorubicin exposure in Huh7 hepatoma cells results in enhanced cytotoxicity and cellular oxidative stress: implications for acute and chronic care of obese cancer patients†
Abstract
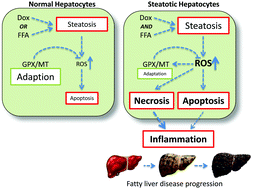
There has been a dramatic increase in the number of clinically obese individuals in the last twenty years. This has resulted in an increasingly common scenario where obese individuals are treated for other diseases, including cancer. Here, we examine interactions between lipid-induced steatosis and doxorubicin treatment in the human hepatoma cell line Huh7. The response of cells to either doxorubicin, lipid-loading or a combination were examined at the global level by DNA microarray, and for specific endpoints of cytotoxicity, lipid-loading, reactive oxygen species, anti-oxidant response systems, and apoptosis. Both doxorubicin and lipid-loading caused a significant accumulation of lipid within Huh7 cells, with the combination resulting in an additive accumulation. In contrast, cytotoxicity was synergistic for the combination compared to the individual components, suggesting an enhanced sensitivity of lipid-loaded cells to the acute hepatotoxic effects of doxorubicin. We demonstrate that a synergistic increase in reactive oxygen species and deregulation of protective anti-oxidant systems, most notably metallothionein expression, underlies this effect. Transcriptome analysis confirms synergistic changes at the global level, and is consistent with enhanced pro-inflammatory signalling in steatotic cells challenged with doxorubicin. Such effects are consistent with a potentiation of progression along the fatty liver disease spectrum. This suggests that treatment of obese individuals with doxorubicin may increase the risk of both acute (i.e. hepatotoxicity) and chronic (i.e. progress of fatty liver disease) adverse effects. This work highlights the need for more study in the growing therapeutic area to develop risk mitigation strategies.

 Please wait while we load your content...
Please wait while we load your content...