Synthesis and photophysical characterization of dimethylamine-derived Zn(ii)phthalocyanines: exploring their potential as selective chemosensors for trinitrophenol†
Abstract
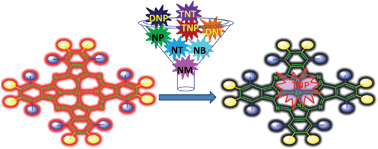
We report a novel synthetic approach, with good yields, for the synthesis of selective dimethylamine-substituted phthalonitriles (1–3) in the presence of triethyl phosphite and dimethylformamide at 160 °C. The peripherally modified dimethylamine substituted Zn(II)phthalocyanines (ZnPc1–3) with varied numbers and positions of dimethylamine groups were prepared for a systematic investigation of the effect of the substituents on their electronic and spectroscopic properties. Compounds show strong aggregation behaviour in methanol and this behaviour decreases with the increase of the alkyl chain length of the alcohol solvents (i.e., from methanol to octanol). The fluorescence quantum yields of ZnPc1–3 showed an excellent correlation with the extent of the molecular aggregation. The versatility of the ZnPc1–5 compounds possessing both electron donating and electron withdrawing substituents at their periphery is investigated towards the detection of nitroaromatic compounds (NACs) in solution and in the vapour phase. It was found that ZnPc1–5 exhibit high selectivity towards trinitrophenol (TNP). A good linearity of fluorescence detection using ZnPc3 as the fluorescent probe was observed in the concentration range of 50 × 10−6 to 450 × 10−6 M in chloroform, with a detection limit (LOD) of 11 ± 2 ppm. Stern–Volmer (SV) and DFT studies reveal that the fluorescence quenching behaviour occurs through photo-induced electron transfer from the excited state of ZnPcs to TNP with static quenching behaviour occurring in a predominant fashion. The formation of a porous morphology of ZnPc3 thin films promotes high selectivity and accessibility to TNP vapours (7.7 × 10−3 ppb).

 Please wait while we load your content...
Please wait while we load your content...