Engineering aligned electrospun PLLA microfibers with nano-porous surface nanotopography for modulating the responses of vascular smooth muscle cells
Abstract
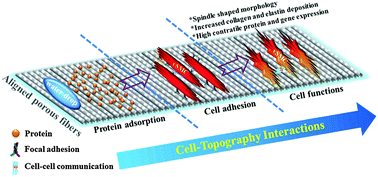
In tissue engineering research, aligned electrospun ultrafine fibers have been shown to regulate cellular alignment and relevant functional expression, but the imposed effect of individual fiber surface nanotopography on cell behaviour has not been examined closely. This work investigates the impact of superimposing a nano-pore feature atop individual fiber surfaces on the responsive behaviour of human vascular smooth muscle cells (vSMCs) for blood vessel tissue engineering. Well-aligned ultrafine poly(L-lactic acid) (PLLA) microfibers with an average fiber diameter of ca. 1.6 μm were fabricated by using a novel stable jet electrospinning (SJES) method. Ellipse-shaped nano-pores with varied aspect ratios (defined as long-to-short axis ratio) of 2.7–3.9, corresponding to a surface nano-roughness in the range of 54.8–110.0 nm, were in situ generated onto individual fiber surfaces by varying ambient humidity from 45% to 75% during the SJES process. The presence of elliptical nano-pores on fiber surfaces affected the characteristic anisotropic wettability of the aligned PLLA fibers and contributed to greater protein adsorption (up to 17.59 μg mg−1). A 7 day in vitro assessment of human umbilical arterial SMCs cultured on these aligned nano-porous fiber substrates indicated that cellular responses were in close correlation with the elliptical nano-pore feature. A pronounced fiber surface nanotopography was superior in soliciting favorable cellular responses, leading to enhanced cell attachment, proliferation, alignment, expression of the vascular matrix proteins and maintenance of a contractile phenotype. This study thus suggests that introduction of an elliptical nano-pore feature to the aligned microfiber surfaces could provide additional dimensionality of topographical cues to modulate the vSMC responses when using the aligned electrospun ultrafine fibers for engineering vascular constructs.

 Please wait while we load your content...
Please wait while we load your content...