Insight into quinoxaline containing D–π–A dyes for dye-sensitized solar cells with cobalt and iodine based electrolytes: the effect of π-bridge on the HOMO energy level and photovoltaic performance†
Abstract
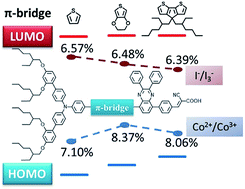
Three new quinoxaline-based organic dyes (AQ201, AQ202, and AQ203), containing thiophene, 3,4-ethylenedioxythiophene (EDOT), and cyclopentadithiophene (CPDT) in the π-system, respectively, have been designed and synthesized for dye-sensitized solar cells. Different from the traditional donor–π-bridge–acceptor (D–π–A) type dyes, the dissymmetric π-bridge on both sides of quinoxaline enables great flexibility in fine-tuning the absorption spectra and energy levels of the resultant molecules. By changing the π-bridge between the bulky triphenylamine donor and quinoxaline group, a negative shift was observed regarding the highest occupied molecular orbital (HOMO) levels of AQ201, AQ202, and AQ203 dyes (0.88, 0.79, and 0.72 V vs. NHE, respectively), while the lowest unoccupied molecular orbital (LUMO) levels of these dyes remained the same (−1.19, −1.20, and −1.20 V vs. NHE, respectively), which, in turn, resulted in a gradual shift of the absorption spectra of AQ dyes. The absorption spectra properties of the dyes are also analysed by density functional theory. The calculated results in combination with the experiments indicate that the absorption bands are mainly dominated by charge transfer transitions from the HOMO and HOMO−1 orbital to the LUMO. In all cases, the [Co(bpy)3]2+/3+ redox-shuttle afforded superior solar cell performance compared to I−/I3−. More importantly, dye AQ202 shows the highest power conversion efficiency (PCE) of 8.37% with the [Co(bpy)3]2+/3+ based electrolyte by maintaining a balance between the spectral absorption range and driving force for dye regeneration. Transient photocurrent decay experiments as well as electrochemical impedance spectroscopy indicate that the lower HOMO levels lead to higher electron lifetime and dye regeneration efficiency.

 Please wait while we load your content...
Please wait while we load your content...