Integrated molecular, morphological and interfacial engineering towards highly efficient and stable solution-processed small molecule solar cells†
Abstract
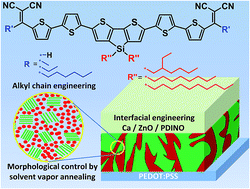
The synthesis of a series of A–π–D–π–A oligomers bearing coplanar electron-donating dithieno[3,2-b:2′,3′-d]silole (DTS) unit linked through bithiophene π-bridges with the electron-withdrawing alkyldicyanovinyl (alkyl-DCV) groups is described. This study demonstrates a systematic investigation of structure–property relationships in this type of oligomer and shows obvious benefits of alkyl-DCV groups as compared to the commonly used DCV ones, in terms of elaboration of high performance organic solar cells (OSCs). Considerable efforts have been made to improve the power conversion efficiency (PCE) of oligomer-based OSCs by diverse strategies including fine-tuning of the oligomer properties via variation of their terminal and central alkyl chains, blend morphology control via solvent vapor annealing (SVA) treatment, and surface modification via interfacial engineering. These efforts allowed achieving PCEs of up to 6.4% for DTS(Oct)2-(2T-DCV-Me)2 blended with PC70BM. Further morphological investigations demonstrated that the usage of SVA treatment indeed effectively results in increased absorption and ordering of the BHJ composite, with the only exception for the most soluble oligomer DTS(Oct)2-(2T-DCV-Hex)2. Besides, a detailed study analyzed the charge transport properties and recombination loss mechanisms for these oligomers. This study not only revealed the importance of integrated alkyl chain engineering on gaining morphological control for high performance OSCs, but also exhibited a clear correlation between molecular ordering and charge carrier mobility respective to carrier dynamics. These results outline a detailed strategy towards a rather complete characterization and optimization methodology for organic photovoltaic devices, thereby paving the way for researchers to easily find the performance parameters adapted for widespread applications.

 Please wait while we load your content...
Please wait while we load your content...