Catalytic effect of two-phase intergrowth and coexistence CuO–CeO2
Abstract
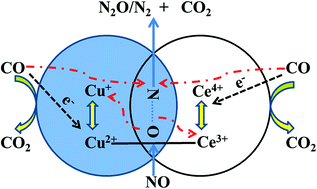
Intergrowth CuO–CeO2 oxides were synthesized using an improved amorphous citric precursor (IACP) method. In order to explore the relationship between structure modulation and catalytic performance, samples were characterized using XRD, XPS, SEM, EDX, HRTEM, O2-TPD, TG-DSC, OSC, H2-TPR, and in situ DRIFTS measurements. Catalytic activities were then investigated in CO + O2 and NO + CO model reactions. Results indicate that the CuO–CeO2 samples form an intergrowth system which is made up of imperfect monoclinic crystal CuO and cubic crystal CeO2. Strong interactions between CuO and CeO2 occurred in the intergrowth process, making the Cu species of CuO gradually form Cu+ and produce corresponding oxygen vacancies with the increase of CeO2 content, which results in an imperfect [Cu1−x2+Cux+][O1−x/2□x/2] structure and improvement of the redox ability of CuO. Meanwhile, the CeO2 retains a few Ce3+ and corresponding oxygen vacancies. As a result, the OSC and the catalytic performance of the samples are greatly enhanced. These results are partly due to the intergrowth and coexistence effects of CuO–CeO2.

 Please wait while we load your content...
Please wait while we load your content...